Crude oil in its natural form is of little commercial use. But when it is separated or broken down into separate parts, it becomes one of the most valuable natural resource in world. Crude oil is a mixture of hydrocarbon compounds with a wide range of boiling points from 100° F (38° Celsius) to 1400° F (760° Celsius).
Crude oil is separated in different parts using process called Distillation. In this process process, liquid mixture is partially vaporized. The vapors are then condensed, separating the individual components of the mixture.
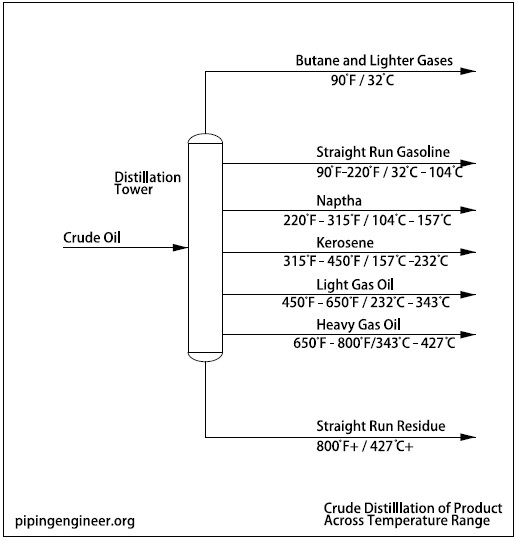
As the temperature of crude oil is raised, the initial boiling point (IBP) is reached. As boiling continues, the temperature rises. The lightest material, butane, is produced first, at IBP, just below 100° F (38° C); the heavier materials are produced below 800° F(427° C).
The residue includes everything above 800° F (427° C).
Following figure shows the distribution of different products at the various temperature ranges.
The evolution of distillation tower is best explained in three basic steps:
 | 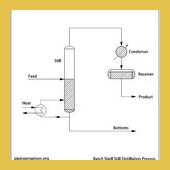 | |
| In the batch shell still process, the still is partially filled with a set feed …Read More.. | ||
 | 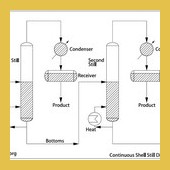 | |
| In the continuous shell still process, several shell stills are linked in series to form a …Read More.. | ||
 | 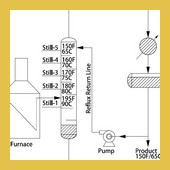 | |
| Similar to continuous shell still, the fractional distillation process is made up of several stills linked …Read More.. | ||

